In the dynamic field of AI technology, a pressing challenge for the drug discovery (DD) community, particularly in structural biology and computational chemistry, is the creation of innovative models finely tuned for drug design . The main challenge lies in accurately and efficiently predicting molecular properties crucial for understanding protein-ligand interactions and optimizing binding affinities, essential for advancing effective drug development initiatives.
In today's field of structural biology and drug design, researchers typically rely on existing data sets and methods, which have inherent limitations such as structural inaccuracies, crystallographic artifacts, and difficulties in accurately capturing the dynamic nature of protein-ligand interactions. Traditional approaches to predicting molecular properties often lack the necessary details for complex protein-ligand interactions, neglecting the vital role of dynamics and flexibility in understanding binding mechanisms and affinity.
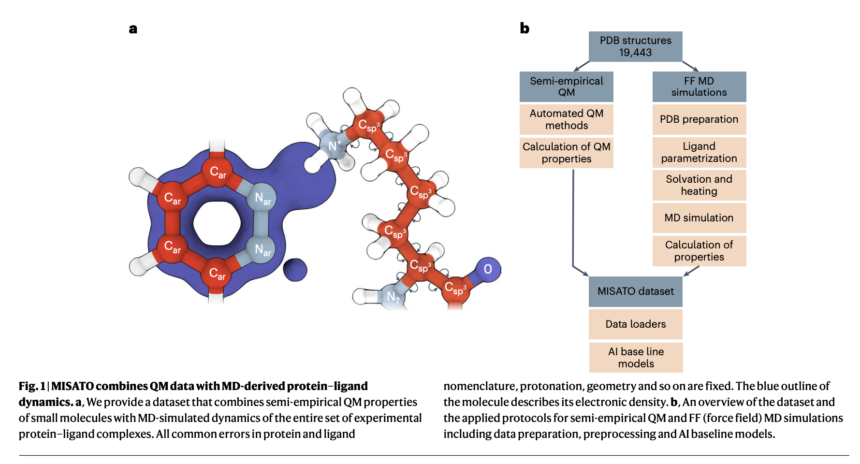
Researchers from the Institute for Structural Biology, the Technical University of Munich, the Jülich Supercomputing Center, Helmholtz AI, the University of Cambridge, the Jagiellonian University and the Institute for Computational Biology propose MISATO, marking a transformative shift in drug discovery and structural biology methodologies. MISATO addresses the limitations of existing methods by integrating ligand data refined by quantum chemistry, molecular dynamics (MD) simulations, and advanced AI models. This comprehensive approach facilitates a nuanced understanding of molecular properties, capturing details of electronic structure and dynamic behavior crucial for accurate predictions.
MISATO takes a holistic approach, using semi-empirical quantum chemistry methods to refine ligand data sets. This method captures electronic properties with high precision, while also analyzing details of electronic structure and dynamic behavior, essential for accurate predictions. Additionally, classical MD simulations within MISATO characterize the dynamic behavior and conformational landscape of protein-ligand complexes, providing insight into binding mechanisms and flexibility. MISATO's built-in AI models, such as graph neural networks (GNN), are trained on this enriched dataset to predict properties such as adaptability, binding affinities, and thermodynamic parameters. Extensive experimental validations confirm the effectiveness of these models in accurately predicting key molecular properties crucial for drug discovery.
In conclusion, MISATO represents a major advancement in AI-based drug discovery and structural biology. By integrating quantum chemistry, MD simulations and advanced AI models, MISATO provides a holistic and robust solution to the challenges of structure-based drug design, improving precision and efficiency and equipping researchers with powerful tools .
Check Paper. All credit for this research goes to the researchers of this project. Also don’t forget to follow us on Twitter. Join our Telegram channel, Discord ChannelAnd LinkedIn Groops.
If you like our work, you will love our bulletin..
Don't forget to join our 42,000+ ML subreddit